Unproven Stem Cell Interventions: How Texas Can Promote Patient Safety and Education
Table of Contents
Author(s)
Kirstin R.W. Matthews
Fellow in Science and Technology PolicyAkshaya Venkatesh
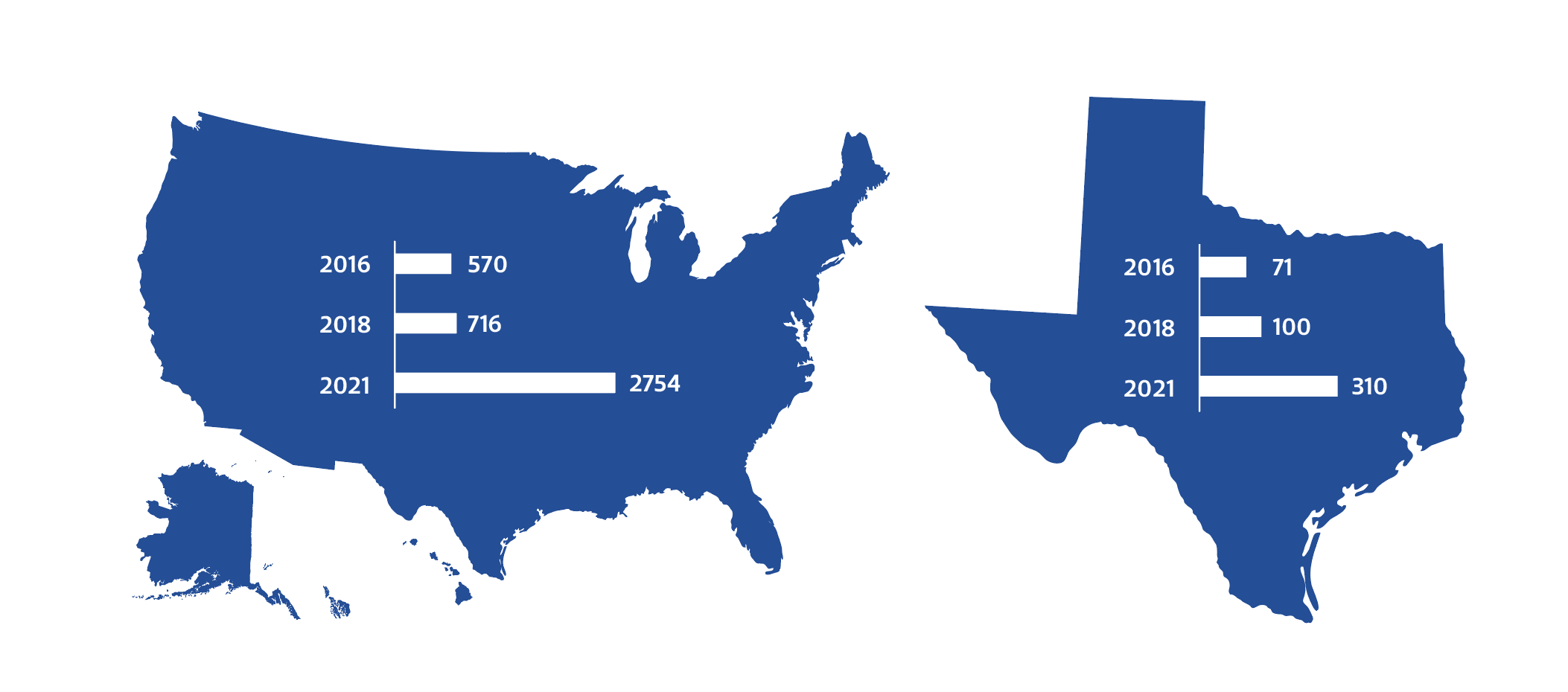
Graduate Student Intern, Center for Health and BiosciencesSince 2016, the number of stem cell clinics in Texas has grown by 336%.[1]
The Risks and Benefits of Stem Cell Interventions
Stem cell interventions (SCIs) are an exciting area of research that shows promise for the field of regenerative medicine. But most SCIs are still experimental — meaning they lack safety and effectiveness data and regulatory approval from the U.S. Food and Drug Administration (FDA) — and many clinics in Texas use misleading tactics to advertise unproven SCIs for a variety of conditions. Patients who undergo these therapies without proper education risk experiencing side effects like sepsis, blindness and even death.
Recommendations to Improve Patient Safety
To improve patient safety and education surrounding SCIs, we recommend that Texas policymakers require clinics and physicians offering SCIs to:
- Display disclaimers for experimental SCIs.
- Place disclaimers on advertisements for experimental SCIs.
- Report adverse events related to SCI treatments to Texas medical boards.
Download the handout for more details, and read the full policy brief on stem cell intervention policy measures here.
Notes
[1] Leigh Turner, “The US Direct-to-Consumer Marketplace for Autologous Stem Cell Interventions,” Perspectives in Biology and Medicine 61, no. 1 (2018): 7–24, https://doi.org/10.1353/pbm.2018.002; Leigh Turner, “The American Stem Cell Sell in 2021: U.S. Businesses Selling Unlicensed and Unproven Stem Cell Interventions,” Cell Stem Cell 28, no. 11 (2021): 1891–95, https://doi.org/10.1016/j.stem.2021.10.008; and Paul S. Knoepfler and Leigh Turner, “The FDA and the US Direct-to-Consumer Marketplace for Stem Cell Interventions: A Temporal Analysis,” Regenerative Medicine 13, no. 1 (2018): 19–27, https://doi.org/10.2217/rme-2017-0115.
This material may be quoted or reproduced without prior permission, provided appropriate credit is given to the author and Rice University’s Baker Institute for Public Policy. The views expressed herein are those of the individual author(s), and do not necessarily represent the views of Rice University’s Baker Institute for Public Policy.


